Pharmacists and antimicrobial stewardship
Many people may find it surprising that pharmacists work in many other pharmacy fields other than behind the counter at a retail pharmacy. There are clinical pharmacists who work in different specialties. Infectious disease pharmacists (ID pharmacists) are among one of the specialties. They are experts in determining which antimicrobial drugs and doses should be used in the treatment of patients with infections.
We are both pharmacy students that have an interest in becoming ID pharmacists after graduation. Here we share our perspectives on the concept of phage therapy as pharmacy students.
What is an infectious disease pharmacist?
ID pharmacists mainly work in hospitals, clinics, and may have academic research positions in universities. Typically they are not hired in retail pharmacies, and perform clinical duties rather than filling and verifying prescription orders.
Pharmacists who do not go through additional residency or fellowship training tend to work in retail pharmacies and hospital pharmacist positions that require little to no clinical roles. Pharmacists that complete residencies gain additional disease management training in multiple clinical specialties, learn to conduct clinical research, and give pharmacotherapy advice to physicians to optimize drug selection and dosing.
A pharmacist who then chooses to complete a second fellowship would choose a specialty, such as infectious disease. Infectious disease pharmacy residencies expose pharmacists for a whole year in infectious disease clinical settings so that they can learn to master medication therapy management in complex ways and perform specialty research. Due to the additional training required for pharmacists to participate in antimicrobial stewardship, these programs are mainly seen in hospitals and clinics rather than in retail pharmacies.
Retail pharmacists and antibiotic stewardship
Retail pharmacists mainly fill, verify, and consult patients on their medications. Even though most retail pharmacists do not have antimicrobial stewardship training, they still have basic knowledge on the antibiotic resistance crisis. They can choose to not fill a prescription without first speaking to a patient’s physician if an antibiotic is inappropriate for the patient’s disease state or if it is the wrong dosage for the patient’s height, weight, and other factors.
Christine’s Perspective
There is starting to be more of an increasing need for infectious disease pharmacists. Many doctors are giving antibiotics unnecessarily for colds, cases of the flu, and coughs. ID Pharmacists in the United States help minimize the inappropriate use of antimicrobial drugs by educating physicians and creating antimicrobial stewardship programs that improve patient outcomes for patients who have infections by reducing the chance of a patient’s body becoming resistant to the drug that they are taking for their infection.
Selecting a dose for an antimicrobial drug can be difficult for pharmacists. However, ID pharmacists tend to use pharmacokinetic/pharmacodynamic (PK/PD) profiling of antibiotics to determine the best dose. Pharmacists may also recommend alternative anti-infective therapies other than antibiotics.
I am a pharmacy student who has worked in both hospital and retail pharmacies in the U.S. As an intern filling antibiotics in a hospital pharmacy, I have seen many cases in which antibiotics do not always cure the diseases that they are used for. Infections such as Staphylococcus aureus have a high rate of multi-drug resistance. There are many antibiotics given to patients conjunctively that are not always effective. This has prompted many clinicians and pharmacists to look into new treatment options.
Yasmine’s Perspective
In Morocco, where I am studying pharmacy, biological pharmacists have a vital role in antimicrobial resistance control. They are involved in many stages of antibiotic therapy. Before setting up a patient’s treatment, a clinical sample is taken to determine the bacteria responsible for the infection, and then an antibiogram is done. These pharmacists must ensure the outcome of the patient through biological monitoring. The biological pharmacist then recommends the right choice of antibiotics to the physician in charge.
With the development of phage therapy and through the use of diagnostic tools, pharmacists can take part in the fight against antibiotic resistance by studying in their laboratory the possible associations of phages with antibiotics and finding which duos work for each bacterial infection.
Phage-antibiotic synergy
Yasmine’s Perspective
The rapid emergence of antibiotic resistance led scientists to approach novel strategies to control bacterial infections and limit the rise of multi-drug resistant organisms. Combined phage-antibiotic therapies are a promising therapeutic approach, and multiple research articles have shown encouraging results with this synergistic combination against bacteria.
It has been shown that sub-lethal doses of antibiotics can lead to higher burst-size and thus phage-mediated bacterial decline. This phenomenon termed as phage-antibiotic synergy or PAS has been first introduced by Comeau et al. (2007), involving a uropathogenic strain of E. coli and a lytic siphovirus. In fact, using lower doses of antibiotics may stimulate the production of phages, and accelerate the lysis of infected cells, hence allowing phages to spread more rapidly.
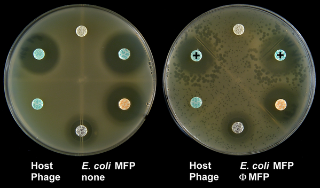
The PAS effect of phage ΦMFP on E. coli. The left-hand control plate shows the sensitivity of the E. coli strain to different antibiotics. On the right plate where phages were added, disks containing β-lactam antibiotics aztreonam and cefixime (indicated by “+” symbols) produced large phage plaques in their proximity. Image reproduced from Comeau et al. (2007) with permission.
Christine’s Perspective
Infectious disease pharmacists who conduct bench-top research may further study phage-antibiotic combinations. Although there are not many pharmacists interested in this research focus, Yasmine and I are interested in researching more about this once we get additional training, since there are many unanswered questions about this synergy. What I find fascinating is that studies show that a multi-drug resistant organism will become resistant towards the antibiotic or the phage, but not both at the same time, leading to the eventual death of the resistant organism.
Some questions I want to answer are:
- Which multi-drug resistant organisms will these synergies be most effective in killing?
- What are the doses needed for each combination?
- Do phages ever do the work better alone?
- Will these combinations be effective at killing the organisms even with long term use?
Targeting biofilm infections
Yasmine’s Perspective
Due to their low susceptibility to antibiotics, biofilms are frequently involved in many chronic infections. PAS can also target biofilms because of the ability of certain phages to penetrate these protective structures. Phages can produce enzymes such as depolymerases and endolysins, that can expedite the breakdown of extracellular matrices of biofilms, thus making bacteria accessible to antibiotics and phages.
The clinical exploitation of PAS would have multiple benefits, such as limiting the amount of antibiotics used, allowing bacteria to regain susceptibility to antibiotics, and managing the emergence of multi-drug resistant infections. However, much work still needs to be done to know if this synergistic combination would work with all antibiotics and all phages, and identify which duos actually work.
Conclusion
In conclusion, pharmacists have a promising role in assisting with the formation and maintenance of new antimicrobial therapies. Although we need a deeper understanding of phage-antibiotic synergy mechanisms, in vitro studies have shown good results, which should encourage scientists and pharmacists to continue on this path.
Our future prospects
Christine
My goal after graduation is to become an academic ID pharmacist. I would like to research antibiotic/phage combinations for disease states that have the highest rates of antibiotic resistance. Hopefully after discovering new antibiotic/phage combinations and phage cocktails, I’ll be able to use them translationally, especially in healing diseases such as cholera in third world countries that have no access to antibiotics. I also want to teach my findings to other students, and assist in ending the antimicrobial resistance crisis once and for all.
Yasmine
My goal once I have my PharmD is to become a pharmacist specialized in infectious diseases. There’s still a lot of research ahead in this field. I aspire to learn more about phage therapy, and contribute to reducing the spread of antibiotic resistance. The use of phage therapy has to be standardized and there needs to be a concerted effort to promote education on phage therapy. Overall, I hope to move up and have a great impact.
Further reading
This article was kindly edited by Lizzie Richardson, our volunteer guest editor. Thanks Lizzie!







